Electroplating gives aluminum alloy parts a strong and attractive finish; but bubbling can ruin the surface. This is a common occurrence but can be avoided with proper preparation and process control. In this blog post we will explain the main reasons for bubbling. We will also show you how to get a smooth and bubble free finish.
Causes of Bubbling During Electroplating of Aluminum Alloy

Electroplating protects aluminum and gives it a clean finish. Sometimes though, bubbles appear during the process and mess up the result. This section looks at the main reasons why bubbles form.
1. Hydrogen Evolution and Gas Entrapment
A chemical reaction at the cathode surface can release hydrogen gas during electroplating. These gas bubbles can stick to the aluminum and prevent the metal coating from spreading evenly. This creates tiny pits.
Also if gas stays trapped under the new layer, it can expand later, especially when heating or drying. The expansion builds pressure and pushes the plating outward. This creates big blisters on the part.
2. Surface Contamination and Poor Cleaning
A clean surface is essential for the plating to stick well. Any leftover grease, oils or polishing compounds can cause big problems. These contaminants block the new metal layer from bonding with the aluminum. Ultimately weak spots and gaps form under the plating which can cause bubbles or make the coating peel off completely.
At RICHCONN, we use automated ultrasonic degreasing and clean‐room assembly to assure every part is spotless before plating.
3. Micro Porosity in Substrate
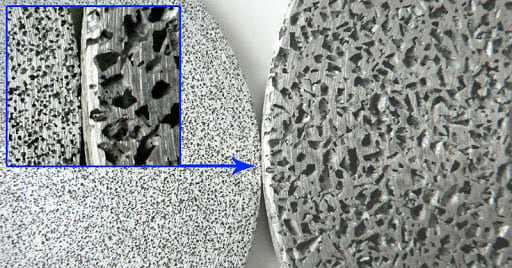
Tiny voids or pores, called micro‐porosity, exist inside die‐cast aluminum parts. These microscopic cavities form naturally during the casting procedure. They can trap gases or cleaning solutions within the metal. When electroplating or heating happens, the trapped substances expand and push against the plated layer. This pressure can lift the coating and create blisters on the surface.
4. Bath Chemistry and Operating Conditions
Bath chemistry and operating conditions of electroplating bath directly affect the coating’s quality. If the bath mixture is incorrect or the electrical current density is set too high, hydrogen bubbles can form faster. Moreover inadequate agitation fails to remove these bubbles from the part’s surface. When bubbles are trapped in the plating, they leave pits and other imperfections behind.
5. Thermal Expansion and Post‐Plating Baking Effects
Drying or baking steps cause any gases or moisture under the plating to expand. As the pressure builds up under the coating, blisters can form. Small changes in temperature can make gases expand big time. Post‐plating heat treatment control is key to prevent bubbling and get a smooth finish.
Specific Bubbling Defects
After knowing what causes bubbling, now you can look at the main bubbling defects that occur during aluminum electroplating.
Blistering
Gas trapped under the coating can expand and create blister. This defect usually forms in areas with pores or weak spots in the aluminum. As a result, air filled bubbles rise to the surface which ultimately weakens the coating. Eventually the finish suffers and the part is more prone to corrosion.
Pitting

Pitting shows up as groups of tiny holes on the surface. When hydrogen bubbles stay on the part during plating, they prevent even metal deposition. These pits make the part look worse and also reduce its strength and protective function.
Prevention of Bubbling in Aluminum Electroplating

To stop bubbling, focus on proper preparation and strict process control. Here are the steps you can take to address the most common causes of bubbling.
1. Thorough Cleaning and Surface Preparation
A clean surface is key to good plating results as contaminants lead to poor adhesion. Therefore start with a detailed cleaning routine that uses alkaline degreasing and acid pickling to remove oxides and oils. Use a “water‐break” test to check for cleanliness—a smooth film of rinse water means the surface is clean. Keep the time between cleaning and plating as short as possible so the aluminum doesn’t form new oxides.
2. Mitigating Hydrogen Impact
Hydrogen gas causes most of the pitting seen in electroplated aluminum. To control this, use pulse or interrupted current plating. These methods let gas escape before it becomes trapped. Lowering the current density also reduces how fast hydrogen forms. For added protection, add anti-pitting additives or wetting agents to the bath. These chemicals stop bubbles from sticking to the surface which leads to a smoother finish.
3. Optimize Bath Conditions and Agitation
Bath conditions must be stable for a smooth coating. Keep the temperature steady because changes can cause gas buildup. Make sure the chemical balance supports metal deposition. Also use mechanical or air‐sparged agitation to move hydrogen bubbles away from the part’s surface. This stops pitting and creates a uniform high quality layer.
RICHCONN uses real‐time IoT monitoring to detect any pH or current changes early. This assures every part meets quality standards.
4. Substrate Inspection and Pore Treatment
Problems mostly start with the aluminum itself especially in die‐cast alloys that have micro‐porosity. These small pores can trap cleaning fluids and gases which later cause blisters under the plating. Use X‐ray or ultrasonic inspection to find these defects. Once a defect is found, seal the problematic area. You can also use pre‐plating steps like acid etching, baking or vibration to force out trapped gases. Moreover these treatments also give the coating a solid base.
5. Post‐Plating Treatments
Quality control doesn’t stop after the plating bath. A post‐plating bake or hydrogen bake‐out lets trapped gases escape before blisters can form. This step is normally done at 375°F for 4 hours and should be done within an hour of plating. This process protects the coating.
Finish with a thorough rinse to remove any leftover chemicals. This final rinse helps stabilize the finish and how well it sticks over time.
Summary Table
| Problem | Prevention Strategy |
|---|---|
| Hydrogen gas formation | Optimize current, add anti‐pit agents, agitate |
| Surface oxide / re‐oxidation | Thorough cleaning, acid activation |
| Porosity & trapped air | Pre‐bake, vacuum rinse, pore sealants |
| Oils, salts & solvents | Multi‐step cleaning, careful rinsing |
| Poor plating conditions (current, temp) | Continuous monitoring, stable bath setup |
| Thermal expansion post‐plating | Controlled baking & drying procedures |
RICHCONN uses a full range of prevention methods, from cleaning steps to post‐plating processes. This way aluminum parts remain bubble free and meet high standards.
To Sum Up
Bubbles after electroplating aluminum alloys come from a number of factors like surface debris, trapped gases as well as tiny pores in the material. Managing the bath composition, cleaning procedure and post‐plating steps reduces defect rates and improves the finish.
If you need professional plating services or flawless CNC machined aluminum parts then contact Richconn. We’re here to help 24/7.
Related Questions
Yes. Some alloys, especially die cast alloys, tend to develop more pores and blisters. These flaws can hold gas and plating liquid which makes bubbles more likely to form.
It depends on your requirements. Anodizing builds up the aluminum’s own oxide layer which increases wear and corrosion resistance. Electroplating adds a separate metal layer which can improve heat transfer or provide a decorative look.
Yes. Anti‐pitting agents reduce the surface tension of the plating solution. This change lets hydrogen bubbles escape from the metal surface therefore they don’t cause pits or coating defects.
Hydrogen porosity leaves small gaps inside the aluminum casting. When electroplating is done, these gaps trap hydrogen gas and solution. Bubbles can then form under the plated layer which weakens how well it sticks